Novel Biomarker for Personalizing Cancer
University of Pittsburgh researchers have identified a type-I interferon response and exhaustion gene signature biomarker to predict response to immune checkpoint inhibitor (ICI) therapies. Through analysis of transcriptional and proteomic dynamics of CD8+ tumor-infiltrating lymphocytes (TIL) at a clonal level, it is possible to personalize ICI treatment for patients with optimized therapeutic responses, reduced risk of unnecessary treatment and improved patient outcomes.
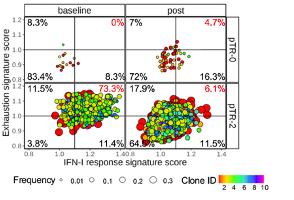
Description
ICI regimens targeting the PD-1/PD-L1 pathway have revolutionized cancer therapy by ensuring the patient’s immune system can recognize and destroy cancer cells. However, not all patients have responded equally well and recent research has suggested that different ICI therapies act on distinct biological pathways. Through the identification of 10-gene type-I interferon and exhaustion gene signatures in CD8+ TIL it could be possible to identify those patients most likely to respond to specific combination ICI therapy. Conversely, those patients who do not express this unique gene signature may be better suited to other ICI regimens, saving time and costs to the patient while reducing the risk of unnecessary and potentially harmful treatment.Applications
• Head and neck squamous cell carcinomas (HNSCC)• Oropharyngeal cancer
• Optimizing immune checkpoint therapy
Advantages
Current immunotherapeutic approaches broadly target ICI. However, recent research has demonstrated that different ICI regimens may act through distinct pathways. This specific gene signature has the potential to serve as a biomarker for determining which ICI combinations are most likely to yield a positive response in individual patients. Screening patients for this biomarker could result in the development of personalized therapies and more efficient treatment.Invention Readiness
As part of a clinical trial comparing neoadjuvant anti-PD-1 (nivolumab), anti-PD-1+CTLA-4 (ipilimumab) and anti-PD-1+LAG-3 (relatlimab) therapies in treatment naïve, stage II-IVa locally advanced resectable HNSCC patients, the transcriptional and proteomic dynamics of TIL were characterized before and after treatment. Single cell RNA sequencing revealed a gene signature associated with increased response to anti-PD-1+anti-LAG-3 therapy. Patients exhibiting a type-I interferon response and exhaustion gene signature in CD8+ TIL, CD4+ conventional and regulatory T cells, and NK cells, exhibited a more favorable response to anti-PD-1+anti-LAG-3 therapy. The absence of this gene signature could serve as a biomarker to identify patients already exhibiting LAG-3 and PD-L1 positivity most likely to benefit from anti-PD-1+anti-LAG-3 therapy, as well as patients who may benefit from other forms of immunotherapy.IP Status
Patent PendingRelated Publication(s)
Li, H., Zandberg, D. P., Kulkarni, A., Chiosea, S. I., Santos, P. M., Isett, B. R., Joy, M., Sica, G. L., Contrera, K. J., Tatsuoka, C. M., Brand, M., Duvvuri, U., Kim, S., Kubik, M., Sridharan, S., Tu, F., Chen, J., Bruno, T. C., Vignali, D. A. A., & Cillo, A. R. (2025). Distinct CD8+ T cell dynamics associate with response to neoadjuvant cancer immunotherapies. Cancer Cell, 0(0). https://doi.org/10.1016/j.ccell.2025.02.026
